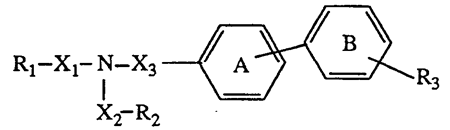
The company governed by the laws of Switzerland, Novartis AG, is the holder of patent EP 0 443 983 entitled “Acyl compounds”, whose subject-matter is a group of antihypertensive compounds, including valsartan, pharmaceutical preparations containing them and processes for the preparation of these compounds.
 ,
,
This patent, filed on 12 February 1991, was to have expired on 12 February 2011. However, Novartis AG endeavoured to extend that protection by obtaining the grant of the supplementary protection certificate (SPC) No. 97 C 0050. This SPC should normally expire on 13 May 2011 but its validity was extended until 13 November 2011, through a “paediatric extension”.
The company governed by the laws of France, Novartis Pharma, was the holder of an exclusive licence under the French designation of patent EP 0 443 983 and of an exclusive licence under SPC No. 97 C 0050. It markets in France two pharmaceutical products containing valsartan, for which it is the holder of different MAs, under the TAREG and COTAREG trademarks. TAREG is indicated for the treatment of high blood pressure, heart failure and post-myocardial infarction. COTAREG is a medicinal product for human use, which combines two active ingredients, valsartan and a diuretic, hydrochlorothiazide “HCTZ”, and which is indicated for the treatment of high blood pressure.
The company governed by the laws of Iceland, Actavis Group PTC EHF, obtained in France, on 30 November 2009, the grant of MAs for pharmaceutical products which challenge COTAREG: “valsartan hydrochlorothiazide Actavis 80mg/12.5mg” and “valsartan hydrochlorothiazide Actavis 160mg/25mg”. It designated Actavis France as the exploiting company of these marketing authorisations.
The Novartis companies, after having advised the Actavis companies of the existence of their SPC rights until 13 May 2011 and until 13 November 2011 because of the paediatric extension, served a summons dated 11 January 2011 on the Actavis companies to a preliminary hearing at set times.
Pursuant to Article L. 615-3 of the French Intellectual Property Code, which implements Article 9 of Directive 2004/48/EC and notably makes it possible to request an interlocutory injunction intended to “prevent any imminent infringement” of patents and related rights (SPC), the Novartis companies asserted the “threat of an imminent infringement” of their rights (patent EP 0 443 983 and SPC No. 97 C 0050) on the grounds of the different actions for injunction in Germany and in Great Britain and above all, the steps made in France to market, as of May 2011, the pharmaceutical products including valsartan, for which the defendants obtained MAs and a reimbursement rate by the CEPS. According to the claimants, the French designation of patent EP 0 443 983 and the SPC No. 97 C 0050, covering valsartan, could be asserted against any product containing valsartan, including a product containing valsartan and another product as a diuretic such as hydrochlorothiazide. On that basis, they requested that the Judge, firstly, enjoin the defendants, under penalty, from manufacturing, importing, marketing, using and holding pharmaceutical preparations implementing the features covered by patent EP 0 443 983 and SPC No. 97 C 0050, secondly, order the publication of the entire decision to be handed down on the home page of the Actavis companies’ website.
The Actavis companies responded that the pharmaceutical products comprising valsartan alone, on the one hand, and valsartan combined with hydrochlorothiazide, on the other hand, constitute different products and that SPC No. 97 C 0050, which received a paediatric extension, only covers the use of valsartan alone; that the SPC does not confer the same rights to the holder as the patent; that this SPC and its paediatric extension, which are connected to an MA, cannot be asserted to prohibit medicinal products covered by another MA, resulting from a combination such as valsartan and a diuretic.
All the parties seem to implicitly admit that a patent covering an active ingredient such as valsartan may be asserted against the manufacturing and marketing of medicinal products comprising valsartan alone or valsartan combined with other active ingredients, as long as this combination does not deprive valsartan of its specific pharmaceutical activity claimed in the patent.
However, the parties disagree on the effect of the SPC. The claimants conceive the SPC as nothing more than the continuation of the patent and of its exclusive rights on the product covered by the MA, valsartan, so that the SPC may be asserted, like the patent, against the manufacturing and marketing of medicinal products comprising valsartan alone or valsartan combined with other active ingredients.
The defendants, on the contrary, insist on the link that exists between the SPC and a specific MA covering a specific product, in order to restrict the scope of the SPC to the sole product covered by the specific MA, thereby excluding from the scope of the SPC the medicinal products which are the subject-matter of a different MA, even if they include valsartan among their active ingredients. They conclude that the SPC does not confer the same rights as the patent and may only be asserted against the manufacturing or marketing of medicinal products comprising this product alone, i.e. in the present case, valsartan, and not of medicinal products combining this product with another product since each of these medicinal products is necessarily the subject-matter of a different MA other than the specific MA to which the SPC is linked.
The legal question submitted to the Judge was clear and well-known, as it had already been submitted to other courts in Europe. In the interpretation of Regulation No. 469/2009/EC concerning the supplementary protection certificate for medicinal products, shall the emphasis be placed on Article 5 (“Subject to the provisions of Article 4, the certificate shall confer the same rights as conferred by the basic patent and shall be subject to the same limitations and the same obligations”) or Article 4 (“Within the limits of the protection conferred by the basic patent, the protection conferred by a certificate shall extend only to the product covered by the authorisation to place the corresponding medicinal product on the market and for any use of the product as a medicinal product that has been authorised before the expiry of the certificate”)?
In the order handed down on 28 January 2011, the Presiding Judge of the Tribunal de Grande Instance of Paris expressly pronounced himself in favour of the interpretation proposed by the claimants (see also Tribunal de Grande Instance of Paris, 12 February 2010, Docket No. 10/51453, “Another SPC Coming”).
He interprets Article 4 of Regulation No. 469/2009, pursuant to its title (“Subject-matter of the protection”), as a mere reminder of the fact that the subject-matter of the protection of a SPC does not necessarily coincide with the whole subject-matter of the basic patent. Indeed, firstly, the subject-matter of the protection of an SPC shall only be a “product” (i.e. the active ingredient or combination of active ingredients of a medicinal product) protected by the basic patent. However, the subject-matter of a basic patent may be far larger than this simple “product”. The subject-matter of the basic patent may be, for example, not only a product but a group of products, as in the present case where it was “a group of antihypertensive compounds” of which valsartan, the subject-matter of the SPC, was only one of the patented compounds. The subject-matter of the basic patent may also be very different from a simple product, as in the present case where the patent also covered “processes for the preparation of [the antihypertensive] compounds”. Secondly, the subject-matter of the protection of an SPC shall only be a product protected by a basic patent and for which, as a medicinal product, a marketing authorisation has been obtained. This analysis allows the Judge to conclude that Article 4 can be read as follows: “Within the limits of the protection conferred by the basic patent [patent EP 0 443 983], the protection conferred by a certificate [SPC No. 97 C 0050] shall extend only to [the active ingredient, i.e., valsartan] covered by the authorisation to place the corresponding medicinal product on the market and for any use of the product as a medicinal product that has been authorised before the expiry of the certificate.” The SPC No. 97 C 0050 protects only valsartan, for the treatments specified in the MA, i.e. for the treatment of high blood pressure, heart failure and post-myocardial infarction; it protects no other active ingredient disclosed in patent EP 0 443 983 nor any treatment other than those specified in the MA.
With the subject-matter of the protection of the SPC having been so precisely delimited pursuant to Article 4 of Regulation No. 469/2009, the Judge then applied Article 5 of the same regulation to underline the fact that the SPC confers on its subject-matter “the same rights as conferred by the basic patent”. Consequently, the Novartis companies, enjoying the same rights as conferred by the basic patent, “can oppose any use of this active ingredient for treating high blood pressure, alone or in combination with another active ingredient”.
With the obvious nature of the infringement, which can occur following the introduction of pharmaceutical products “valsartan hydrochlorothiazide Actavis 80mg/12.5mg” and “valsartan hydrochlorothiazide Actavis 160mg/25mg” on the market before 13 November 2011, thus established, the Judge, pursuant to Article L. 615-3 of the French Intellectual Property Code, enjoined the defendants from offering for sale and selling, that is to say, marketing, these pharmaceutical preparations before 13 November 2011, under a €100 penalty per tablet offered for sale and sold, in bulk or in any other packaging form, with the penalty taking effect as of the day of the order.
The Judge thus restricted this preliminary injunction to only the marketing acts of the presumably infringing pharmaceutical preparations, deciding on the contrary that “the manufacture, holding and import of generic drugs before the end of the protection period of the patent do not constitute an act of infringement”.
However, such a restriction seems to have no legal basis. Article L. 613-3 a) of the French Intellectual Property Code does include the manufacture, holding or import within the scope of the exclusive right of the patentee and consequently within the scope of the infringement when these acts are committed without the patentee’s authorisation. Even if Article L. 613-5 d) of the French Intellectual Property Code provides that the rights conferred by the patent do not allow its holder to oppose “the studies and assays required to obtain a market authorisation for a medicine, as well as the acts necessary to their completion and for obtaining the market authorisation”, it seems rather difficult to presume that any act of manufacture, holding or import would coincide with an act necessary to the completion of the studies and assays required to obtain a market authorisation.
Lastly, however, the Judge dismissed the claim for a judicial publication on Actavis’ website on the grounds that the irreparable nature of such a measure prohibits its pronouncement in preliminary proceedings.
Original French decision.
English translation.
Author: Nicolas Bouche, Head Legal Research and Literature, Véron & Associés, Paris, France
________________________
To make sure you do not miss out on regular updates from the Kluwer Patent Blog, please subscribe here.



One comment
Comments are closed.