On 15 March 2011, in a dispute opposing the companies E.I Du Pont de Nemours and Merck and companies Mylan and Qualimed, the Cour d’Appel of Paris confirmed the order handed down on 12 February 2010 by the President of the Tribunal de Grande Instance of Paris, subject-matter of a previous post.
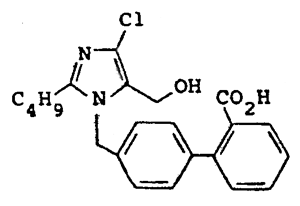
The American company E.I. Du Pont de Nemours and Company (hereinafter referred to as “Du Pont”) has two European patents relating to anti-hypertensive compounds: imidazoles acting as angiotensin II receptor blockers. The first patent covers a group of anti-hypertensive imidazoles, among which losartan, pharmaceutical compositions containing them, as well as processes for the preparation of those compounds. The second patent covers pharmaceutical compositions combining anti-hypertensive imidazoles and diuretics (which, by eliminating part of the water and sodium contained in blood, also contribute to lowering pressure level by decreasing blood volume) as well as processes for the manufacturing of those compositions.
On the basis of those two European patents, Du Pont also obtained two supplementary protection certificates (SPC) in accordance with Regulation (EEC) No. 1768/92 of 18 June 1992 concerning the creation of a supplementary protection certificate for medicinal products (replaced, in the meantime, by Regulation (EC) No. 469/2009 of 6 May 2009). Those two SPCs came into force on the respective expiry dates of the two patents (on 9 July 2007 and 5 January 2009). The first SPC covered losartan while the second covered losartan in combination with hydrochlorothiazide (HCTZ), a diuretic.
As the first SPC was to expire on 2 September 2009, Du Pont applied for and obtained a paediatric extension on the basis of Article 36 of Regulation (EC) No. 1901/2006 of 12 December 2006 on medicinal products for paediatric use. The validity of the first SPC was thus extended by 6 months to 2 March 2010.
However, the company did not apply for such an extension for the second SPC which was to expire on 15 February 2010.
When the generic manufacturer Mylan obtained, by way of AFSSAPS decisions on 5 June 2009, the registration of its generic drugs Losartan HCTZ Mylan 50mg and 100mg which combine the active ingredients losartan and hydrochlorothiazide, Du Pont then warned Mylan that putting those generic drugs on the market before the end of the first SPC extension, that is, before 2 March 2010, would amount to infringement.
Mylan replied that it nonetheless intended to commercialise its generic drugs as of 15 February 2010, the expiry date of the second SPC, which was the only one to cover the combination of losartan and hydrochlorothiazide at issue.
On 2 February 2010, Du Pont and its exclusive licensee (Merck) initiated preliminary proceedings against Mylan and Qualimed with an emergency motion to be heard on very short notice, to enjoin Mylan and Qualimed, under penalty, from manufacturing, having manufactured, importing, offering for sale and selling, using and holding pharmaceutical compositions reproducing the characteristics covered by the claims of the first patent and of the first SPC. The summons was served on 2 February 2010 and the hearing took place on 5 February 2010.
In its order handed down on 12 February 2010, the Presiding Judge of the Tribunal de Grande Instance of Paris decided that, although the generic drug combining losartan and a diuretic is the subject-matter of the second SPC, which covers precisely such combination of active ingredients, only until its expiry date (on 15 February 2010), this generic drug, insofar as it contains losartan acting as the active ingredient against hypertension, also falls under the subject-matter of the first SPC, which covers losartan alone, until its respective expiry (on 2 March 2010). It then enjoined Mylan and Qualimed, under penalty, from offering for sale and selling, using and holding pharmaceutical compositions reproducing the characteristics covered by the claims of the first patent and of the first SPC.
Mylan and Qualimed then lodged an appeal. They requested that the judge hold that they did not commit any act of infringement by placing their drug Losartan HCTZ on the market on 15 February 2010 and requested that the judge find that the requirements for obtaining a paediatric extension of the duration of the protection provided by SPC No. 95C0018 as laid down in Article 36 of Regulation (EC) No. 1901/2006 were not met.
The Cour d’Appel firstly underlined that the procedure provided by Article L. 615-3 of the French Intellectual Property Code (IPC) was independent and distinct, especially in its conditions of application, from the common procedure of référé as laid down in Articles 808 and 809 of the French Code of Civil Procedure (CPC).
The court therefore contradicted the order handed down on 12 February 2010 by the President of the Tribunal de Grande Instance of Paris which had linked Article L. 615-3 IPC and Article 809 CPC together. After having cited the provisions of Article L. 615-3, the Judge in preliminary proceedings had concluded that the case was referred to him “under the same conditions as those provided in Article 809 of the French Code of Civil Procedure which allows the Judge in preliminary proceedings, even when confronted with serious objections, to order protective measures or measures to keep the status quo of the matters as required, either to protect from an impending damage or to abate a manifestly illegal nuisance”. The judge had also mentioned the concept of “impending damage” further along in the decision.
The rigour of the Cour d’Appel must be approved since it corresponds to the requirements of Article 810 CPC which states that “The powers of the president of the Tribunal de Grande Instance as provided for under the aforegoing two Articles shall extend to all matters where summary interlocutory proceedings cannot be instituted”. Article 810 CPC requires a contrari to abandon any reference to Articles 808 and 809 CPC as soon as a particular procedure of référé exists in a particular matter. Such is the case here of patent law and related titles (SPC, certificates of utility) where Article L. 615-3 IPC provides for such a special procedure which makes it possible to obtain from the Judge in preliminary proceedings measures to prevent the imminent infringement of the rights conferred by the intellectual property or to prevent the continuation of acts alleged to be counterfeiting.
The court then validated the paediatric extension of SPC No. 95C0018. Mylan and Qualimed disputed the validity of this paediatric extension claiming that, contrary to the requirement of Article 36 of Regulation (EC) No. 1901/2006, Du Pont did not demonstrate that all the paediatric marketing authorizations (MAs) had been granted in the 27 States of the European Union at the time of the filing of the application for the paediatric extension. The court rejected this interpretation of the requirements of Article 36 of Regulation (EC) No. 1901/2006. It recalled that pursuant to Article 36, the application for an extension includes the result of the studies conducted according to an approved paediatric investigation plan and the six-month extension of the period may be granted only if the product is authorised in all the Member States. It decided that what the text actually requires is that the product have obtained the initials MAs and not the paediatric MAs in all the Member States. In support of this solution, it was already argued that Article 36 refers only to the fact that the product is authorised and does not specify that when submitting the application, all pediatric AMM must have been obtained. It also argued that “the extension of the period of protection granted in this context is a reward for carrying out paediatric studies but does not mean that the product is necessarily efficient in the paediatric population and that the extension of the period of protection may be granted although the paediatric indication is not authorised” (Article 36 paragraph 1, second sentence). It added a practical consideration by noting that “to request that the 27 paediatric MAs be supplied on the date of the filing of the application is not possible in practice in the absence of a centralised procedure of registration and owing to the variable speed at which agencies grant these authorisations”. Finally, it noted that this position was finally adopted and implemented by the INPI (Institut National de la Propriété Industrielle – French Industrial Property Office).
SPC No. 95C0018 being thus valid until 2 March 2010, the Cour d’Appel concluded that the generic drugs which Mylan and Qualimed intended to place on the market could constitute, should this commercialisation take place before 2 March 2010, an infringement of the product protected by SPC No. 95C0018 since they contained losartan. In doing so, the Cour d’Appel rightly confirmed the reasoning of the Judge in preliminary proceedings in the order handed down on 12 February 2010. The generic drugs of Mylan and Qualimed are indeed products which combine losartan and a diuretic and this combination is the subject-matter of SPC No. 98C0025, expired on 15 February 2010. However, since these generic drugs contain and use losartan as the anti-hypertensive active ingredient, they do indeed constitute an infringement of the SPC No. 95C0018 which covers only losartan and expires on 2 March 2010.
In the present case, the basic patent EP 0 253 310 covers a group of products, among which losartan, as anti-hypertensive drugs and the SPC No. 95C0018 specifically covers losartan which is the product covered by the MA. Pursuant to Article 5 of Regulation (EC) No. 469/2009 (which replaces Regulation (EEC) No. 1768/92), the certificate shall confer on the product covered by the MA the same rights as those conferred by the basic patent. A generic drug which contains losartan among its active ingredients and which is used as an anti-hypertensive drug necessarily constitutes, until 2 March 2010, an infringement of the SPC since “infringement is assessed through similarities and not merely through differences, so that characteristics added to the characteristics claimed by a patent do not eliminate the infringement”.
The Cour d’Appel finally wondered whether the requirement of Article L. 615-3 IPC, i.e. the claimant shall make likely that an infringement of its rights is imminent, had been met in the present case. It defined the imminent infringement as “an infringement which has not yet been committed but which is bound to happen, should the current situation continue”. And in the assessment of the circumstances of the present case, the court first noted that certain circumstances were not probative of an imminent infringement. Legislation authorises generic manufacturers to undertake all the necessary formalities to place their products on the market before the extinguishment of the industrial property rights over the proprietary drugs. The generic manufacturers may also have their products registered in the list of refundable medicinal products and in the index of generic drugs. These formalities may be completed subject to the notification of the right holder. Consequently, the steps taken by Mylan and Qualimed before the expiry of the rights of Du Pont and Merck were not sufficient to demonstrate the imminent infringement of the latter’s rights. The court also adds that neither the simple warning letter sent by the right holders to the appellants, nor Mylan and Qualimed’s reply disputing the analysis of the rights of Du Pont and Merck is probative.
The court then noted that no evidence of imminent infringement was provided, i.e. elements relating to the set up of any manufacturing, door-to-door selling or advertisement, such as preparations for manufacturing or marketing, for distributing brochures or advertisements announcing the products’ launch, or the visit of medical doctors in view of the prescription of these generic drugs.
However, the court still recognized that the imminence of the infringement was demonstrated by Mylan and Qualimed’s admission which, in their own pleading, claimed that they did not market their medicinal products, did not make preparations for any marketing before 15 February 2010 and did not commit acts of infringement by placing on the market their medicinal products on 15 February 2010.
Consequently, the court affirmed the order in that it prohibits the marketing of Losartan HCTZ 50mg and Losartan HCTZ 100mg until 2 March 2010 (refusing however to pronounce a penalty). On this issue, moreover, it did not give complete satisfaction to Du Pont and Merck which requested that the interlocutory injunction be extended to the acts of manufacturing, importing, using and holding any pharmaceutical product containing losartan reproducing claims 1 to 5 of patent EP 0 253 310 and of SPC No. 95C0018 until 2 March 2010. The court explained this refusal noting that the medicinal products Losartan HCTZ 50mg and 100mg were “generic drugs which were granted the required authorisations from the public authorities and which should be marketed as of the end of the protection conferred by the patent and the SPC held by” Du Pont and Merck, that “only marketing before expiry of the protection may be considered to be an infringement”.
In other words, the court inferred from the various legal provisions according to which the generic manufacturers are authorised to take all the necessary formalities to place their products on the market before the extinguishment of the industrial property rights over the proprietary drugs (Article L. 5121-10 §1 of the French Public Health Code), the idea that the generic manufacturers shall be able to market their generic drugs as of the end of the protection conferred by the patent and the SPC (Article L. 5121-10 §3 of the French Public Health Code). And consequently the acts of manufacturing, importing, using and holding made before the expiry of these rights but only in expectation of a marketing after the expiry of the protection conferred by the rights, may not be prohibited.
Such reasoning is however questionable since these acts of manufacturing, importing, using and holding are acts of infringement of the patent (pursuant to Article L. 613-3 IPC) or of the SPC (pursuant to Article 5 of Regulation (EC) No. 469/2009). The court, by refusing to consider and treat these acts as infringing acts on the grounds that they are the necessary prior to the marketing of the generic drugs after the expiry of the patent or of the SPC, created a new limitation, not provided by a legal text, to the rights conferred by the patent or the SPC. However, only the law and not the judge himself may create such limitations to the rights conferred by the patent or the SPC. Article L. 5121-10 of the French Public Health Code effectively authorises the grant of a MA before the expiry of the intellectual property rights (patent, SPC) covering the proprietary medicine. The acts made by the generic manufacturer and which are necessary to the progress of the administrative procedure of the grant of the MA, even before the expiry of the intellectual property rights, shall certainly be removed from the list of infringing acts. But this limitation, as with any other, must be strictly interpreted and only the acts strictly necessary to the grant of the MA shall be removed from the list of infringing acts. Consequently, the acts of manufacturing, importing, using and holding, made before the expiry of the rights, not with the aim of obtaining the MA but rather with the aim of preparing the marketing after the expiry of the rights, cannot benefit from this limitation.
This analysis is moreover confirmed in French patent law by Article L. 613-5 d) which provides as limitation to the rights conferred by the patent only “the studies and assays required to obtain a market authorisation for a medicine, as well as the acts necessary for their completion and for obtaining market authorisation”. It clearly means a contrario that the acts which serve only to prepare future marketing do not benefit from the limitation and remain acts of infringement.
Original French decision.
English translation.
Author: Nicolas Bouche, Head Legal Research and Literature, Véron & Associés, Paris, France
________________________
To make sure you do not miss out on regular updates from the Kluwer Patent Blog, please subscribe here.


